Medical Injection Molding
Elimold provides a range of medical injection molding manufacturing services to global medical product manufacturers, including plastic injection molding and injection mold manufacturing. Our medical injection molding facilities follow strict design protocols and process controls to ensure that all products meet the required specifications. Depending on customer needs and product requirements, we can provide manual or fully automated production lines for molding and assembly. Contact us now for a free instant quote.
Our Medical Injection Molding Services
Elimold has the innovative medical product manufacturing injection molding solutions and experienced services your healthcare organization needs to turn your medical device product ideas into reality. We provide automated medical device manufacturing solutions, no matter how complex. From rapid prototyping to mass injection molding production solutions, our capabilities can meet your unique manufacturing needs.
Together with our partners, we develop high-precision medical injection molding products and components with expertise, flexibility and the highest quality standards. We ensure smooth industrialization with professional project management. Our strong product development and quality assurance programs ensure that medical products have the highest reliability in application, excellent quality and meet strict industry standards.

In-House Medical Product Injection Molding Facility Capabilities
Our 55,000 square foot manufacturing facility in Shenzhen, China is dedicated to precision injection molding of medical device and diagnostic components. With 130 injection molding machines ranging from 28 to 2500 tons, we can handle small, complex parts as well as large components and housings. With full ISO 13485 certification and FDA registration, we meet the most stringent medical device manufacturing regulations. Our cleanroom injection molding environment provides extremely low particle levels, which is critical for manufacturing implantable devices, surgical instruments, IVD components, and drug delivery systems. We can meet the tight tolerances and validations required for Class III and Class II medical hardware. Our expertise in medical molding processes and rigorous quality systems enable us to produce millions of components each year for industry-leading medical OEM.


Product Design and Development Support
Elimold continuously invests in engineering resources. We have the expertise, skills, and technical knowledge to help you through all stages and levels of complexity of medical plastic device injection molding manufacturing. Our manufacturing experience and investment in the early product design phase helps our customers save resources and time. By collaborating early in the product design phase development, we can eliminate common errors and mitigate redesign issues that may arise due to the complexity of the product, so we encourage medical device manufacturers to work with us during the product design phase. We can also help you establish the entire assembly and test procedures, and all established procedures are optimized to provide the best quality and most efficient turnaround time. In addition, we also help maintain the documentation required for medical device molding.
Our Turnkey Cleanroom Solutions
Partnering with Elimold means you can take advantage of the benefits of cleanroom manufacturing without having to build a cleanroom yourself. Our certified cleanroom setup improves product quality, minimizes yield loss, and increases efficiency, giving you peace of mind that every part is produced in a sterile, controlled environment.
Elimold’s two Class 100,000/ISO 8 cleanrooms are equipped with the necessary technology and capabilities to ensure that our commitment to quality is uncompromised. Our cleanrooms of over 3,600 square meters include medical device injection molding machines, assembly and sub-assembly cells and machines, and final packaging facilities. The cleanrooms are equipped with the latest technology, such as quality control equipment and robotics, to ensure smooth mass production capabilities. In addition to our production facilities, we have established validation protocols, testing procedures, and process improvement studies to ensure the highest quality and competitiveness. This helps our engineering and manufacturing teams reduce production errors. It also optimizes manufacturing cycles, thereby increasing productivity.

How we maintain cleanroom production conditions
We regularly monitor temperature, humidity, and air filtration to identify and eliminate sources of contamination, ensuring part traceability and compliance. Strict equipment and procedure validation controls ensure our customers’ projects are executed with extreme precision and repeatability. When manufacturing medical devices, medical injection molding companies must maintain a sterile environment. Any bacteria or microorganisms may pose a safety hazard to patients and animals. As a medical device component manufacturer, our investment in a Class 8 cleanroom has resulted in greater efficiency and a better environment, resulting in higher quality medical products at a more competitive price.
Elimold team manages the entire process for you!
Elimold provides top-notch medical injection molding solutions for the growing medical, pharmaceutical and dental industries. We have now expanded our expertise to Class I, II and III medical devices and components using a wide range of medical grade resins. Our state-of-the-art machines, automated quality inspection equipment and science-backed molding processes guarantee that you can rely on our services with confidence. If that’s not enough, we also provide ISO 7 cleanroom molding, assembly and packaging services to provide a complete solution for your medical products. Elimold adheres to the key medical quality standards required for plastic injection molded medical parts through complete quality control and inspection, including IQ, OQ, PQ validation and batch traceability. Trust us to fully handle your medical device injection molding project!

End-to-End Solutions for Medical Device Manufacturers
Design, Development and Proof of Concept
Whether you need concept design, assembly design, DFM or manufacturing design, Elimold’s design team will turn your concept into reality. Once the design is complete, we provide a proof of concept to ensure that the device design concept works properly.
Prototypes & Custom Molding
Whether you have a rough sketch scribbled onto a napkin or a CAD mockup, our engineering team can take your design and create a prototype. We offer other types of molding such as overmolding, insert molding, and LSR molding as well.
Contract Manufacturing
We offer end-to-end contract manufacturing solutions including design, prototyping, ramping up production and automation. Elimold will work with you to develop a plan to scale up production of your product so that the processes can move from partially automated to fully automated as you move to higher volumes.
Packaging, Quality Control and Logistics
Elimold is FDA registered and ISO 13485 certified, and we implement quality control processes for each function to ensure that your products are of the highest quality. Medical devices are sealed in packaging to prevent contamination or exposure to air/moisture.
Our Manufacturing Services
Elimold offers best-in-class medical plastic product manufacturing capabilities, ideal for rapid prototyping and custom production orders. With our own facilities and extensive manufacturing network, we are able to efficiently deliver complex and high-quality medical parts.

Plastic Injection Molding

LSR Injection Molding

Overmolding

Gas Assist Injection Molding

Insert Molding

Structural Foam Molding

Rapid Tooling

Micro Molding

Production Tooling
Medical Plastic Product Injection Molding Prototyping
Medical device prototyping requires the creation of fully functional components for testing in the FDA approval process. Elimold uses state-of-the-art methods and uses the finest medical grade plastic materials to provide medical device prototyping services. We understand the strict regulations required by the healthcare industry. We will take every step to ensure that your prototype meets the Class I, II, or III FDA medical device classification required for your application.


Process Validation Services
In many cases, our medical device customers want to ensure the quality of their parts through process validation. After all, good medical devices depend on Elimold providing high-quality parts every time. We define process validation as the collection and evaluation of data from the product design phase and throughout production to establish scientific evidence that our manufacturing processes can consistently provide quality products. Elimold’s process validation services ensure the production of safe parts, reduce scrap rates, prevent part failures in the field, and ensure that parts meet application, specification and regulatory obligations.
Medical Micromolding Capabilities
Elimold can provide small medical component parts molded to very tight tolerances. The biggest misconception about micro molding is that people think it is just a smaller version of injection molding. The process is completely different, which is why many injection molders have trouble getting into true micro molding with micron tolerances. We have a large team with decades of experience in molding tiny, challenging parts. All of our tooling and automation equipment is designed, built, and maintained in-house 24/7, and our DFM engineers are ready to help you with even the most challenging medical micro molding components.

Comprehensive Medical Injection Molding Solutions
Our approach includes detailed planning and oversight at every stage, ensuring seamless execution and superior quality. We strategically enter the market as a mold maker and precision medical product injection molding manufacturer. We can design the mold around the product and solve your project by considering key factors such as gating, maximum cooling, ventilation, wear and replacement parts, as well as mold life and product volume production. Our project engineering team, each with plastic engineering expertise and experience, can help our customers optimize their product designs for manufacturing, material selection, process optimization and ongoing production support. We support our customers with weekly updates on mold building progress and production approvals. This holistic support system ensures that every project we undertake is successful and focused on innovation, quality and patient safety.
Contact Elimold now for custom medical injection molding
Elimold provides you with medical injection molding solutions, from design to mold to material selection and manufacturing. Contact our professional team now to solve your problems.

In-house Medical Device Mold Manufacturing Facility
At Elimold, we manufacture medical device molds in our own facility, so we have full control over the quality. To design and manufacture the molds, we can follow customer standards and also use our in-house standard designs. Also, we ensure that the molds can be manufactured using imported and certified steels and standard components that you prefer. And this guarantees that we are able to manufacture the best quality medical device molds that medical device manufacturers should adhere to. Elimold’s in-house injection mold manufacturing facility also ensures better lead times and enables us to incorporate any type of minor or simple modifications that the molds may require. Moreover, we also provide expedited services to deliver spare and replacement parts for medical device molds with the shortest lead time. Moreover, we can also guarantee that the quality of the replacement or spare parts meets the quality standards. You will experience fast and direct communication, ultimately leading to a seamless and smooth manufacturing process.
Mold Design Services
Mold design is a critical step in the injection molding process. Once the fit and function of a part has been designed in CAD software, it must be converted into a molding design to ensure that all specified details are captured. In some cases, certain features of a part design may not be manufacturable via the injection molding process. At Elimold, we work with you to achieve the best design for manufacturability. In fact, we have a free guide to help you achieve this.
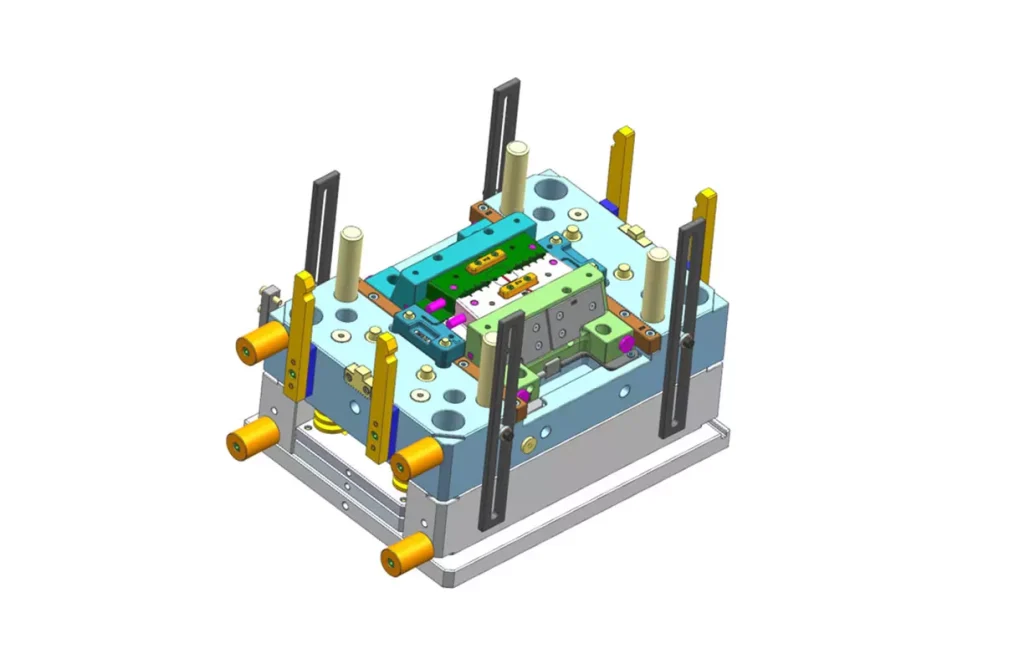
Our Medical Injection Mold Tooling Services
- In-house mold manufacturing
- High-precision machining
- Quick tool iteration and modification
- Quick turnaround prototypes
- Mold flow analysis
- Scientific molding process development
- Single-cavity pilot production injection molds
- High-cavity hot runner mold system


Full integration between tooling, program management and metrology
- Elimold provides information for tool design, supporting decisions on critical areas to insert and retain steel safety.
- Build duplicate metrology fixtures before mold is completed to allow metrology correlation between tool makers and Formula.
- Steel safety recuts can be confidently made at the original tool maker.
- First Article Inspection (FAI) and Capability Testing (CpK) procedures are written to complete R&R measurements before mold is shipped.
- End of Arm Tooling (EoAT) is also manufactured before mold is completed for initial sampling and then shipped with the mold.
- Where applicable, cavity separation equipment is used for production, saving labor and providing easier traceability.
- All mold samples are made on-site by the Formula mold engineer.
Expert Medical Product Assembly Services
Elimold offers tailor-made solutions for your company’s unique manufacturing needs. We meet your quality requirements from the receipt of raw materials to final inspection of finished products. When you work with us, we prioritize safety, quality, and efficiency in all assembly processes to ensure you get the most value from your medical device. With our assembly services, you can rest assured that you will receive innovative products on time with a competitive advantage. Each of our assembly options can be custom to meet your production needs. We follow efficient assembly processes to improve your product quality while saving costs. From proof of concept to steady state production, our expert assembly services include: manual assembly, semi-automated and fully automated, micro-assembly, regular scale assembly, and more.
We are proud to have some of the best facilities in the world. Our facilities are flexible in configuration and equipped with everything we need to provide a wide range of medical manufacturing and assembly services to meet your needs.

Engineering Quality Assurance
At Elimold, our motto is that quality comes first. And quality is an integral part of every project. Our Shenzhen, China facility adheres to the highest standards in medical device manufacturing, maintains ISO 13485 certification and is registered with the FDA. We incorporate our quality management system throughout the product lifecycle, from design and validation to production and post-market surveillance. Our quality engineers can collaborate cross-functionally to ensure compliance every step of the way. Elimold’s custom protocols include installation qualification (IQ), operation qualification (OQ), performance qualification (PQ), and process validation in accordance with ISO 13485. In addition, we can provide full traceability and change control for transparent record keeping. Our metrology laboratory uses advanced measurement equipment for dimensional inspection and functional testing, while using optical comparators, vision systems, and coordinate measuring machines to fully evaluate molded parts and finished device assemblies. We also adhere to strict procedural and documentation disciplines by combining strong quality processes with continuous training. Elimold’s dedication to quality and process control helps reduce risk for our medical device partners.
Ready to get started on your next medical injection molding project?
Our mission is to build strong, long-term relationships with our customers to develop custom plastic manufacturing solutions for your medical device and component needs. When interacting with Elimold, you will always experience one-on-one personalized service and a complete team approach. Whether you are reviewing a quote with our sales engineer representative or discussing a prototype with our team of engineers, you will experience friendly, knowledgeable, and extremely dedicated individuals. We work tirelessly to provide the best value to our customers and are proud of our ability to be flexible to meet your changing needs. Contact us now for a free instant quote.
Comply with FDA Regulations and ISO Standards
When Elimold manufactures injection molded parts and products for our medical industry customers, we consider compliance issues in addition to application and material specific factors. We are well aware that the medical industry is highly regulated. This means that any part or product we manufacture for you, whether through injection molding or other manufacturing processes, will comply with FDA regulations and be ISO certified and meet the appropriate standards.
FDA Regulations
The FDA has strict regulations for the cleanliness and sterility of implantable devices, medical instruments, other medical components, and materials used in cleanrooms. This means you need to ensure that the materials you choose can meet these standards. Therefore, in addition to paying attention to the injection molding process itself, you also need to pass an audit to obtain medical-grade approval.
ISO Certification and Compliance
Elimold ensures that your medical components meet ISO standards. Compliance with ISO 13485:2016 is the standard, but you may also need to meet other standards. In some cases, we can provide compliance to Class I, II, or III requirements or ISO 10993 and other biocompatibility standards.
Medical Grade Injection Molding Material Options
With the most appropriate and advanced thermoplastic and thermoset materials, Elimold helps medical product manufacturers select the right materials – those that provide the specific performance characteristics required for the finished part, product or assembly. Our customers can choose from a wide variety of carefully engineered materials.
- ABS(Acrylonitrile Butadiene Styrene)
- POM(Acetal)
- PMMA(Acrylic)
- LCP(Liquid crystal polymers)
- LSR(Liquid Silicone Rubber)
- PA(Nylon)
- PC(Polycarbonate)
- PBT(Polyester)
- PET(Polyethylene)
- PUR(Polyurethane)
- TPU/TPE(Thermoplastic Polyurethane)
- PEEK(Polyetheretherketone)
- PEI(Ultem)
- PPE+PS(Polyphenylene Ether (Noryl))

Post-processing options
Elimold offers a wide range of post-processing services.
- Hot stamping
- Applying functional/cosmetic coatings such as EMI/RFI shielding or special paints
- Ultrasonic welding and insertion
- Post-Molding Machining
- Silk Screening and Pad Printing
- Paint
- Powder Coating
- Annealing
- Heat Staking
- Ultrasonic Welding & Curing
- Assembly
- Packaging
Medical Injection Molding Finishes
In addition to the surface treatments listed below, Elimold can match other mold textures upon request.
Standard Finish
Moldmaker’s choice of finish. Typically SPI B-3 to D-3 depending on geometry and drafts. Interior, non-cosmetic faces, are typically as-machined.
MoldTech Finishes
Range of finishes including matte, swirls, lines, and patterns. Finishes include: MoldTech – MT11010 MoldTech – MT11020 MoldTech – MT11030 MoldTech – MT112XX MoldTech – MT114XX MoldTech – MT116XX
SPI Finishes
Range of finishes from Grade 3 diamond / high polish to 320 stone low polish. Finishes include: SPI A-1 SPI A-2 SPI A-3 SPI B-1 SPI B-2 SPI B-3 SPI C-1 SPI C-2 SPI C-3 SPI D-1
As Molded
No secondary polishing or grinding. Part will show tooling marks.

Medical Applications of Injection Molding
With injection molding, medical design engineers have the freedom to design complex parts in a variety of materials. The consistency of the plastic injection molding process is ideal for both low-volume and high-volume production, giving your medical device an immediate success.
- Implantables
- Imaging Components
- Ear, Nose & Throat
- Orthopedics
- Regenerative Medical Devices
- Device Housings
- In-Vitro Diagnostics
- Gastroenterology
- Home Healthcare Devices
- Prenatal Care
- Cardiovascular
- Surgical Devices & Instruments
- Pharmaceutical
- Labware
- Dental Products
- Emergency Care
Get a free quote for medical injection molding now
Get a free quote for our custom medical injection molding services now. Whether you have a specific project in mind or need expert guidance on material selection and design, the team at Elimold is here to help.
